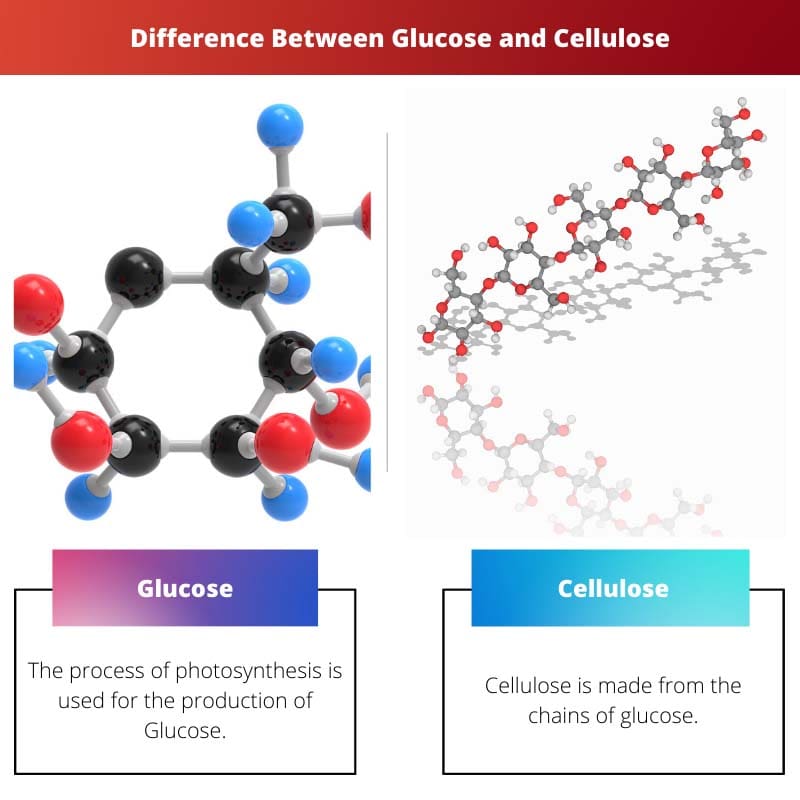
Glucose and cellulose are both carbohydrates composed of glucose monomers. However, their structural arrangement differs significantly. Glucose forms a linear chain, serving as a primary energy source in cells, while cellulose consists of a linear chain of glucose units linked by beta-1,4-glycosidic bonds, creating a rigid and indigestible structure, vital for plant cell walls.
Key Takeaways
- Glucose is a simple sugar used by the body as a source of energy, while cellulose is a complex carbohydrate that makes up the cell walls of plants.
- Glucose is a monosaccharide easily broken down by the body during digestion, while cellulose is a polysaccharide that humans do not easily digest.
- Glucose is found in many foods and is an essential component of the body’s metabolic processes, while cellulose is an important structural component of plant cells.
Glucose vs Cellulose
Glucose is a simple sugar and a form of carbohydrate that is directly absorbed into the bloodstream during digestion and used by the body for energy. Cellulose is a complex carbohydrate and the primary component of plant cell walls, which human digestive enzymes cannot digest.
Glucose is also known as dextrose. It is a part of carbohydrate groups called simple sugars. These simple sugars are called Monosaccharides.
Cellulose-linked D-glucose refers to an organic compound found in Plants. For green plants, it is an important constituent of the primary cell wall structure.
Comparison Table
| Feature | Glucose | Cellulose |
|---|---|---|
| Monomer | Glucose | Glucose |
| Type of Carbohydrate | Monosaccharide (simple sugar) | Polysaccharide (complex sugar) |
| Structure | Single unit of glucose | Long, unbranched chains of glucose linked together by β-1,4 glycosidic bonds |
| Solubility in Water | Highly soluble | Insoluble |
| Digestibility | Easily digestible by humans and provides energy | Not digestible by most humans (provides dietary fiber) |
| Function | Primary source of energy for cells | Main structural component of plant cell walls |
| Examples | Blood sugar, table sugar (sucrose is broken down into glucose and fructose) | Wood, cotton fiber |
What is Glucose?
Glucose is a simple sugar, also known as a monosaccharide, and is a fundamental source of energy for living organisms. Its chemical formula is C6H12O6, reflecting its six carbon atoms, twelve hydrogen atoms, and six oxygen atoms.
Structure of Glucose
- Molecular Structure: Glucose has a hexagonal structure, and each carbon atom is associated with a hydroxyl group (-OH) except for one, which is part of a carbonyl group (C=O).
- Isomerism: Glucose exists in two stereoisomeric forms: D-glucose (dextrose) and L-glucose. However, D-glucose is the biologically significant form.
Sources of Glucose
- Dietary Sources: Commonly found in carbohydrates such as sugars and starches in food, especially in fruits, honey, and carbohydrates from grains.
- Biological Synthesis: Cells can produce glucose through various metabolic pathways, including gluconeogenesis.
Role in Energy Metabolism
- Glycolysis: Glucose is a key substrate in glycolysis, a process that breaks it down into pyruvate, producing adenosine triphosphate (ATP) molecules.
- Cellular Respiration: In aerobic organisms, glucose undergoes cellular respiration, ultimately yielding energy for cellular activities.
Regulation of Blood Glucose
- Insulin and Glucagon: Hormones such as insulin and glucagon play a crucial role in regulating blood glucose levels. Insulin facilitates glucose uptake by cells, while glucagon promotes the release of glucose from the liver.
Medical Significance
- Blood Sugar Levels: Maintaining optimal blood glucose levels is essential for health. Abnormalities, such as hyperglycemia (high blood sugar) or hypoglycemia (low blood sugar), can have serious health implications.
- Diabetes Mellitus: A chronic condition characterized by impaired glucose metabolism, resulting in elevated blood glucose levels. Type 1 and Type 2 diabetes are the two main forms.
Industrial Applications
- Fermentation: Glucose serves as a substrate for fermentation processes, producing ethanol and other valuable products.
- Biotechnology: Used in various biotechnological processes, including the production of biofuels and pharmaceuticals.

What is Cellulose?
Cellulose is a complex carbohydrate and the primary structural component of the cell walls in plants. It is the most abundant organic compound on Earth and plays a crucial role in the biological and industrial realms. This versatile polymer is composed of repeating glucose units linked together by β-1,4-glycosidic bonds.
Structure of Cellulose
The cellulose molecule forms long, linear chains due to the arrangement of glucose units. These chains are held together by hydrogen bonds, creating a strong and stable structure. The linear orientation of cellulose molecules allows them to align and bundle into microfibrils, providing strength and rigidity to plant cell walls.
Biological Significance
- Plant Cell Wall Integrity: Cellulose is a key component of plant cell walls, providing structural support and protection. It contributes to the overall strength and rigidity of plant cells, allowing them to withstand mechanical stress.
- Digestive Resistance in Humans: While humans lack the enzymes necessary to digest cellulose, its presence in dietary fiber is essential for digestive health. Cellulose adds bulk to the diet, aiding in proper bowel function and preventing constipation.
Industrial Applications
- Paper and Pulp Industry: One of the major industrial uses of cellulose is in the production of paper and pulp. The cellulose fibers are extracted from plant sources and processed to create various paper products.
- Textile Industry: Cellulose-based fibers, such as rayon and viscose, are widely used in the textile industry. These fibers are derived from wood pulp and offer a more sustainable alternative to synthetic materials.
Chemical Properties
- Hydrophilic Nature: Cellulose is hydrophilic, meaning it has a strong affinity for water. This property is important in various applications, such as in the pharmaceutical and food industries.
- Chemical Modifications: Cellulose can be chemically modified to enhance its properties. For example, derivatization with certain chemicals can improve solubility, making it suitable for specific applications.
Environmental Impact
- Renewable Resource: As cellulose is derived from plant materials, it is considered a renewable resource. This contributes to its sustainability and reduced environmental impact compared to non-renewable alternatives.
- Biodegradability: Cellulose is biodegradable, meaning it can be broken down by microorganisms. This characteristic is advantageous in waste management and environmental conservation efforts.

Main Differences Between Glucose and Cellulose
- Chemical Structure:
- Glucose: It is a simple sugar and a monosaccharide. Its molecular formula is C6H12O6.
- Cellulose: It is a complex carbohydrate and a polysaccharide. Its molecular formula is (C6H10O5)n, representing a long chain of glucose molecules.
- Monomer vs. Polymer:
- Glucose: It is a monosaccharide, meaning it is a single sugar unit.
- Cellulose: It is a polymer composed of many glucose molecules linked together in a linear fashion.
- Function in Organisms:
- Glucose: It serves as a primary source of energy in many living organisms, participating in cellular respiration to produce ATP.
- Cellulose: It is a structural component in the cell walls of plants, providing rigidity and support to plant cells.
- Digestibility:
- Glucose: It is easily digestible and can be readily used by organisms for energy.
- Cellulose: Most organisms, including humans, lack the enzymes necessary to digest cellulose effectively. It acts as dietary fiber and aids in digestion.
- Bond Type:
- Glucose: It is linked by glycosidic bonds in its various forms.
- Cellulose: Glucose molecules in cellulose are connected by β-1,4-glycosidic bonds, forming a linear chain.
- Solubility:
- Glucose: It is highly soluble in water.
- Cellulose: It is generally insoluble in water due to its extensive hydrogen bonding between glucose chains.
- Location in Organisms:
- Glucose: It is found in various tissues and fluids of organisms, circulating in the bloodstream in animals.
- Cellulose: It is primarily located in the cell walls of plants.
- Energy Storage vs. Structural Role:
- Glucose: It is a source of quick energy storage.
- Cellulose: It provides structural support and is not used as an immediate energy source in most organisms.
- Hydrolysis:
- Glucose: It can be easily hydrolyzed into simpler compounds.
- Cellulose: Hydrolyzing cellulose requires specific enzymes, and its breakdown is more challenging than that of glucose.
- Taste and Texture:
- Glucose: It is sweet in taste.
- Cellulose: It is tasteless and contributes to the fibrous texture of plant-based foods.
- https://www.ahajournals.org/doi/abs/10.1161/01.cir.99.4.578
- https://inis.iaea.org/search/search.aspx?orig_q=RN:17038890

This article discusses the complexities of Glucose and Cellulose, highlighting their importance as energy sources for living beings. The thorough explanation of their manufacturing process and usage provides a deep understanding of how these compounds contribute to the growth and development of plants and their roles in human diets.
The detailed comparison between Glucose and Cellulose makes it clear how they differ in terms of manufacturing process, form, usage, and absorption and digestion. The comparison table makes it easy to recognize and remember these key dissimilarities.
The article provides a comprehensive understanding of Glucose and Cellulose, defining their role in living organisms. Furthermore, it details the main differences between Glucose and Cellulose, emphasizing their importance in the human diet and in the growth of plants.
This article is an excellent resource for understanding Glucose and Cellulose, focusing on their differences and the key role they play in the energy sources for living organisms. The comparison table provides an informative way to recognize and comprehend the contrasting attributes of these compounds.
The detailed explanation of Glucose and Cellulose in this article is valuable for understanding the different sources of energy for human beings. The thorough comparison table aids in recognizing the contrast between these two essential energy sources.
This article provides comprehensive details of the different sources of energy for human beings. In the understanding of Glucose and Cellulose, the article has highlighted the key differences between the two. The comparison table offers an easy-to-understand format to differentiate and understand the two types of energy sources.
This article offers substantial information on the sources of energy for living beings, focusing on the significant differences between Glucose and Cellulose. The content is clear and informative, providing valuable insights into these important energy sources.
The article provides a comprehensive understanding of the contrasting characteristics of Glucose and Cellulose, including the details of their manufacturing process, form, usage, and absorption and digestion. This comparison aids in distinguishing their roles in the human diet and in plant development.
Through detailed information and comparison, the article effectively distinguishes between Glucose and Cellulose as energy sources for living organisms. The explanation of their absorption and digestion clarifies their roles in the diet of humans and in plant physiology.
The article expertly details the differences between Glucose and Cellulose, particularly in relation to their absorption and digestion. The detailed explanation of the manufacturing process and usage of these compounds makes their roles in the diet of humans and growth of plants clear.